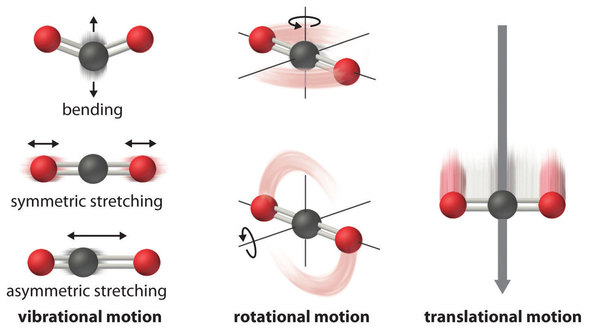
One first demonstrates that zero temperature is unattainable. Entropy denoted by S is a measure of the disorderrandomness in a closed system.
13 6 The Third Law Of Thermodynamics Chemistry Libretexts
Third law of Thermodynamics.

Third law of thermodynamics. This is because a system at zero temperature exists in its ground state so that its entropy is determined only by the degeneracy of the ground state. We may compute the standard entropy change for a process by using standard entropy values for the reactants and products involved in the process. Most thermodynamics calculations use only entropy differences so the.
The Third Law of Thermodynamics The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero. The third law of thermodynamics is essentially a statement about the ability to create an absolute temperature scale for which absolute zero is the point at which the internal energy of a solid is precisely 0. Zeroth law of Thermodynamics.
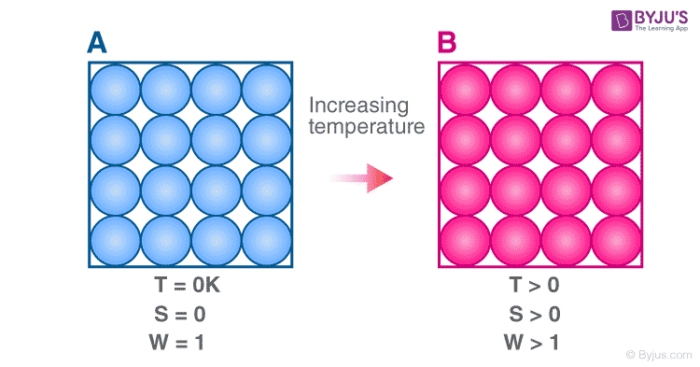
Second law of thermodynamics The entropy of an isolated system not in equilibrium will tend to increase over time approaching a maximum value at equilibrium. This is the lowest point on the Kelvin scale. However if there is even the smallest hint of imperfection in this crystalline structure then there will also be a minimal amount of entropy.
The entropy of a system at absolute zero is typically zero and in all cases is determined only by the number of different ground states it has. The third law of thermodynamics states that the entropy of a system at absolute zero is a well-defined constant. With only one possible microstate the entropy is zero.
A pure perfect crystal is one in which every molecule is identical and the molecular alignment is perfectly even throughout the substance. According to equation 6 𝐶 𝑃tends to approach zero at 0KThis means that at absolute zero the heat capacities of products and reactants in solid state are identical. The entropy of a system at absolute zero is typically zero and in all cases is determined only by the number of different ground states it has.
The third law of thermodynamics establishes the zero for entropy as that of a perfect pure crystalline solid at 0 K. The entropy of a system at absolute zero is typically zero and in all cases is determined only by the number of different ground. Third law of thermodynamics.
Traditionally thermodynamics has recognized three fundamental laws simply named by an ordinal identification the first law the second law and the third law. Google Classroom Facebook Twitter. 1 2 3 A more fundamental statement was later labelled as the zeroth law after the first three laws had been established.
This is the currently selected item. There are three laws of thermodynamics. The third law of thermodynamics states that the entropy of a perfect crystal at a temperature of zero Kelvin absolute zero is equal to zero.
The Third Law of Thermodynamics The Third Law of Thermodynamics says that a perfect crystalline structure at absolute zero temperatures will have zero disorder or entropy. First and Second Laws of Thermodynamics as they apply to biological systems. The third law of thermodynamics predicts the properties of a system and the behavior of entropy in a unique environment known as absolute temperature.
The laws of thermodynamics. The Third Law of Thermodynamics. The Third Law of Thermodynamics The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero.
Second Law of Thermodynamics and entropy. Now let us come back to third law of thermodynamics which says that at absolute zero temperature the entropy of the pure crystal is zero. The change in entropy is equal to the heat absorbed divided by the temperature of the reversible process.
1423 The Third Law of Thermodynamics You Cannot Get Out of the Game The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero. Zeroth law of thermodynamics. First Law of Thermodynamics introduction.
The Third Law of Thermodynamics. The Third Law of Thermodynamics is concerned with the limiting behavior of systems as the temperature approaches absolute zero. Third Law Of Thermodynamics The Third Law states The entropy of a perfect crystal is zero when the temperature of the crystal is equal to absolute zero 0 K The third law of thermodynamics also refers to a state known as absolute zero.
Third Law of Thermodynamics Explained. The Third Law of Thermodynamics means that as the temperature of a system approaches absolute zero its entropy approaches a constant for pure perfect crystals this constant is zero. Second law of thermodynamics.
It states that when two bodies are in equilibrium with a third body then they are also in thermal equilibrium with each other. A pure crystal is the substance in which all the molecules are perfectly identical and the alignment of molecules with each other is perfectly uniform throughout the. Unattainability of Zero Temperature The third law of thermodynamics deals with events as T0 where đQTmight diverge.
The third law of thermodynamic states that as the temperature of a system approaches absolute zero its entropy becomes constant or the change in entropy is zero. First law of thermodynamics. This leads to the suggestion that at absolute zero all substances have the some heat capacity.
Third law of thermodynamics As temperature approaches absolute zero the entropy of a system approaches a constant minimum. The third law of thermodynamics states that the entropy of a system at absolute zero is a well-defined constant.
Third Law Of Thermodynamics Entropy At Absolute Zero


