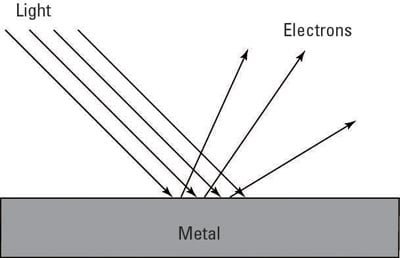
This phenomenon whereby electrons in atoms get liberated by the absorption of energy from incident light is called the photoelectric effect. The emitted electrons are called photoelectrons.

1 Photoelectric Effect 2 Photoelectric Effect What Is It When Metal Surfaces Are Exposed To Electromagnetic Radiation With Sufficient Energy They Absorb Ppt Download
The photoelectric effect is a phenomenon that occurs when light shined onto a metal surface causes the ejection of electrons from that metal.
The photoelectric effect for dummies. The magic that makes solar panels work The photovoltaic effect is the fancy name given to the phenomenon of converting light to electricity in a conventional solar panel. When ultraviolet light is shone on certain metal surfaces electrons are emitted. This is one of the main reasons that scientists chose to treat light as both a wave and a stream of particles.
In a broader definition the radiant energy may be infrared visible or ultraviolet light. Einstein proposed that light consisted of quanta which we call photons. Electrons emitted in this manner may be referred to as photoelectrons.
The photoelectric effect is a phenomenon in physics. In the photoelectric effect electrons are emitted from matter metals and non-metallic solids liquids or gases as a consequence of their absorption of energy from electromagnetic radiation of high frequency short wavelength such as ultraviolet radiation. The effect has found use in electronic devices.
The photoelectric effect refers to what happens when electrons are emitted from a material that has absorbed electromagnetic radiation. The phenomenon is studied in condensed matter physics and solid state and quantum chemistry to draw inferences about the properties of atoms molecules and solids. The photoelectric effect was one of many experimental results that made up a crisis for classical physics around the turn of the 20th century.
The photoelectric effect refers to the emission or ejection of electrons from the surface of generally a metal in response to incident light. At the high photon energies comparable to the electron rest energy of 511 keV Compton scattering may occur pair production may take place at energies over 1022 MeV. Energy contained within the incident light is absorbed by electrons within the metal giving the electrons sufficient energy to be knocked out of that is emitted from the surface of the metal.
I use a fluorescent lamp black light for the ultraviolet light a zinc. The effect is based on the idea that electromagnetic radiation is made of a series of particles called photons. So how do we create a flow of electrons using a solar PV panel and sunshine.
It was the solution to this physics dilemma that catapulted Einstein into prominence in the physics community ultimately earning him the 1921 Nobel Prize. When a photon hits an electron on a metal surface the electron can be emitted. The photoelectric effect was first observed in 1887 by Heinrich Hertz during experiments with a spark gap generator the earliest device that could be called a radio.
Electricity is simply a flow of electrons running around a closed circuit. The photoelectric effect is a property of light that is not explained by the theory that light is a wave. Physicist Albert Einstein was the first to describe the.
The photoelectric effect posed a significant challenge to the study of optics in the latter portion of the 1800s. The photoelectric effect is the emission of electrons when electromagnetic radiation such as light hits a materialElectrons emitted in this manner are called photoelectrons. It was also one of Einsteins first successes and it provides proof of the quantization of light.
These ejected electrons are called photoelectrons. This is due to the fact that when a photon of light incidents on the surface of metal and hits an electron in an atom it contains enough energy to knock the electron out of the atom. In these experiments sparks generated between two small metal spheres in a transmitter induce sparks that jump between between two different metal spheres in a receiver.
It is important to note that the emission of photoelectrons and the kinetic energy of the ejected photoelectrons is dependent on the frequency of the light that is incident on the metals surface. The photoelectric effect occurs with photons having energies from a few electronvolts to over 1 MeV. The photoelectric effect including what is the photoelectric effect and how it works.
It challenged the classical wave theory of light which was the prevailing theory of the time. The effect is often defined as the ejection of electrons from a metal plate when light falls on it. A key experiment that was explained by Einstein using lights particle nature was called the photoelectric effect.
The photoelectric effect is a phenomenon in which electrons are ejected from the surface of a metal when light is incident on it. Photoelectric effect phenomenon in which electrically charged particles are released from or within a material when it absorbs electromagnetic radiation. Photoelectric effect When light shines on a metal surface it emits electrons.
What is the Photoelectric Effect and Why does it Occur.
Photoelectric Effect In Chemistry

Photoelectric Effect Wikipedia
How Physicists Solved The Photoelectric Effect Of Light Dummies