197 79 118. What is the smallest mass in an atom.

Meaning And Representation In Symbols Of Atoms And Sub Atomic Particles Inside Chemistry
What subatomic particle has an approximate mass of 11840.

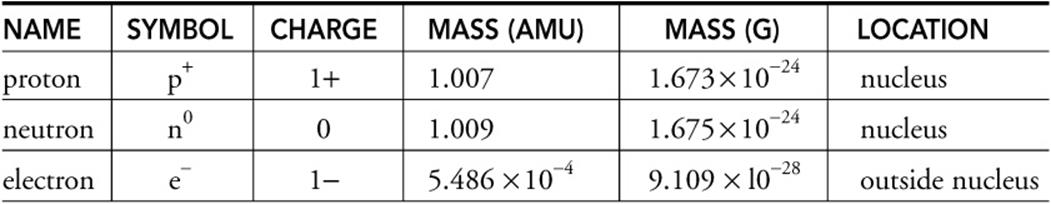
What is the mass of each subatomic particle. For a given element the mass number is the number of protons and neutrons nucleons in the nucleus. The neutron number can be calculated by subtracting the proton mass from the total mass. What is the charge and mass of each subatomic particle.
167493 10 -27 kg slightly larger than that of a proton Electrons. Positive 16726x10-24g neutral 16749 x 10-24g negative 91 x 10-31kg. They have the same atomic number same number of protons and electrons but a different mass number due to more or fewer neutrons.
Each isotope of a given element has the same atomic number but a different mass number A which is the sum of the numbers of protons and neutrons. In the most common isotope of Oxygen the mass is 16 O_816 In this isotope there are also 8 neutrons. The masses of subatomic particles are very tiny.
The proton has a mass of 1007 amu the neutron has a mass of 1 amu. The largest subatomic particle by mass is the proton. The field of subatomic particles has expanded vastly with the construction of powerful particle accelerators for studying the high-energy collisions of electrons protons and the other particles within the matter.
In contrast the electron has a negligible mass of0005 amu. The mass of a proton is approximately 16726 10-24 g. The number of basic subatomic particles is 24 Oxygen because it is number 8 has 8 positive protons.
Because the masses of particles in atoms are so small scientists developed a new unit for them. 79 is its charge atomic number which is both its proton number and electron number. The atom with the smallest mass is the hydrogen atom.
Where is the neutron located. Masses for the three subatomic particles can be expressed in amu atomic mass units or grams. The atomic number of an element is the number of protons in its nucleus.
In this article we will take a look at what are subatomic particles subatomic particles definition and the discovery of. Neutron is an uncharged particle of the nucleus of all atoms EXCEPT hydrogen. What is the charge and mass of each subatomic particle.
Label the diagram below. The negatively charged electron has a mass equal to 1 1837 or 1836 of that of a hydrogen atom. Instead of writing their actual masses in kilograms we often use their relative masses.
The remainder of the hydrogen atoms mass comes from the positively charged proton. What is the charge and mass of each subatomic particle. Subatomic particles include electrons the negatively charged almost massless particles that nevertheless account for most of the size of the atom and they include the heavier building blocks of the small but very dense nucleus of.
The relative masses of atoms are reported using the atomic mass unit amu which is defined as one-twelfth of the mass of one atom of carbon-12 with 6 protons 6 neutrons and 6 electrons. The relative mass of a proton is 1 and a particle with a. Which particle makes the atom an unstable isotope make sure to check the box at the bottom that says stableunstable.
Neutrons are neutral particles having a mass slightly greater than that of the proton. Both neutrons and protons are assigned as having masses of 1 amu each. Atomic Number Atomic Mass and Charge Play with the simulation to identify what each number signifies.
The SI unit known as the atomic mass unit. As a neutral atom the negative electrons must equal the protons so Oxygen also has 8 electrons. Subsequently question is which subatomic particle has the most mass.
Its mass is about 10-27 kg. According to Newtons law all bodies are attracted to each other by a force that depends directly on the mass of each body and inversely on the square of the distance between them. The actual total of subatomic particles is unknown.
Electron-has a -1 charge-Electrical electron-moves fast AROUND center of atom. Subatomic particle also called elementary particle any of various self-contained units of matter or energy that are the fundamental constituents of all matter. Proton-has 1 charge -in center -positive proton.
For simplicity we will use the amu unit for the three subatomics. What is the charge and mass of each subatomic particle Answer The electron has a charge -16 x 10-19 C and a mass 91 x 10-32 kg The proton has a charge 16 x 1019 C and a mass 167 x 10-27 kg The neutron has a charge 0 C it is neutral and a mass 167 x 10-27 kg. The mass of subatomic particles is measured in atomic mass units amu.
Isotope is one of two or more forms of atoms of an element that differ in their number of neutrons. For a pair of masses m1 and m2 a distance r apart the strength of the force F is given by F Gm1m2 r2. The masses of other atoms go up to about 200 times this.
0 although each neutron is made up of charged subatomic particles Mass at rest. - Neutrons will make the atom an unstable isotope when too many are either added or removed. 197 is its mass atomic mass which is the sum of its proton number and neutron number.

Subatomic Particles Atomic Number And Atomic Mass Ppt Video Online Download
What Are The Subatomic Particles In An Atom Including The Charge Location And Mass Of Each And Their Relationship Within The Atom Socratic

The Table Below Gives The Masses Of Two Different Subatomic Particles Found In An Atom Which Of The Brainly Com