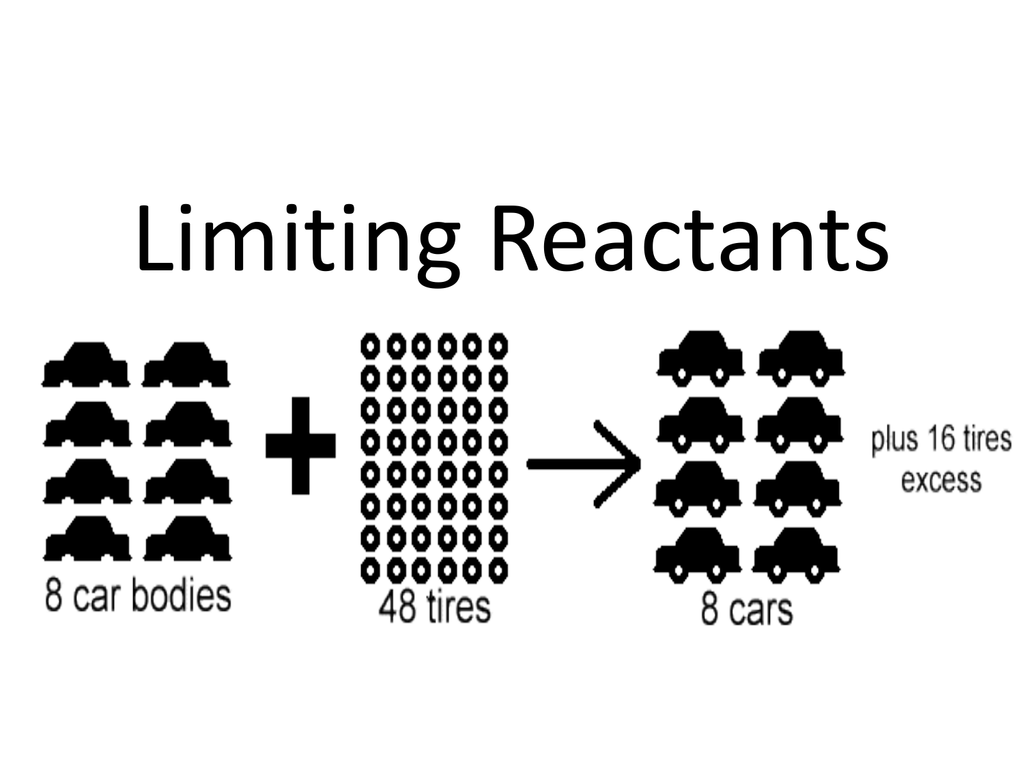
To solve the problem you must identify which of the reactant is going to run out. A chemical reaction involves reactants some reactants in excess and some in limited amounts.
Limiting Reactant And Reaction Yields Article Khan Academy
Let us consider the reaction between solid sodium and chlorine gas.
Limiting and excess reactants. Limiting reactant - limiting reactant is a reactant that determines the amount of product that is formed. The theoretic yield of a reaction is the amount of products produced when the limiting reactant runs out. The limiting reactant of a reaction is the reactant that would run out first if all the reactants were to be reacted together.
The one that is remaining is the excess reactant. If you are given the mass of reactants first convert them to moles and then compare their values to the mole ratio to identify the limiting and excess reactant. So heres the solution.
Given the following equation. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators. The limiting reagent is the reactant which is not present in excess in a reaction.
The reagent that is completely used up or reacted is called the limiting reagent because its quantity limits the amount of products formed. The reactant that is all used up is called the limiting reactant - it sets a limit on how much product can form. A reactant is a compound that is consumed during a chemical reaction.
The reactant that is all used up is called the limiting reactant the reactant that is left over is described as being in excess The mass of product formed in a reaction depends upon the mass of the. 2 determine the limiting reagent b determine the number of moles of carbon dioxide produced c determine the number of grams of H 2O produced d determine the number of grams of excess reagent left 2. The limiting reagent depends on the mole ratio not on the masses of the reactants present.
Difference Between Limiting Reagent and Excess Reagent Definition. Limiting reagent is the reactant of a particular chemical reaction that limits the. Identify the excess reagent.
The COMPLETION is when there will be product formation. The key difference between limiting reactant and excess reactant is that the limiting reactant can limit the amount of final product produced whereas excess reactant has no effect on the amount of final product. Excess reactants on the other hand are the reactants that are still present after the reaction has reached a standstill.
Dont waste good thought. Lets say that youre standing in a queue at your favorite bagel vendor. Once the limiting reactant is completely consumed the reaction would cease to progress.
Determine the limiting reagent if 100 g of ammonia and 100 g of oxygen are present at the beginning of the reaction. Al 2SO 3 3 6 NaOH ----- 3 Na 2SO 3 2 AlOH 3 a If 100 g of Al 2SO 3. This chemistry video tutorial shows you how to identify the limiting reagent and excess reactant.
It shows you how to perform stoichiometric calculations an. Once the limiting reactant gets used up the reaction has to stop and cannot continue and there is extra of the other reactants left over. If you start a reaction with 1 mole of each substance then silver iodide is the limiting reactant and sodium sulfide is the excess reactant.
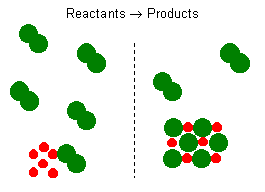
To find the amount of remaining excess reactant subtract the mass of excess reagent consumed from the total mass of excess reagent given. The EXCESS REACTANT is the substance that is excess because of the limiting reactant and the reactantreagent that gives the excess amount in a reaction. Limiting Reagent Before and After Reaction From the illustration shown above it can be observed that the limiting reactant is the reason the reaction cannot continue since there is nothing left to react with the excess reactant.
In a chemical reaction reactants that are not used up when the reaction is finished are called excess reagents. Limiting reactants are those that get completely utilized in a reaction first and thus limit the amount of product that will be produced. To Find the Limiting Reagent.
The reactant that produces a lesser amount of product is the limiting reagent. Limiting reagent is completely consumed during a reaction. Excess reactant - An excess reactant is the amount of reactants present after the required amount of reactants used to make a product.
The mass of product. It is the reactant that entirely consumed over the course of the reaction. Those are called the excess reactants.
The reagent that is used up first is the limiting reactant as it limits the duration and hence the amount of product that a reaction can produce. Before doing anything else you must have a balanced reaction equation. The reactant that is left over is described as being in excess.
Based on your observation come up with a definition for limiting reactant and excess reactant. The limiting reactant or limiting reagent is the first reactant to get used up in a chemical reaction. The reactant that produces a larger amount of product is the excess reagent.

Limiting Reactant General Chemistry I Lab Lecture Slides Docsity
Limiting And Excess Reactants For New Gcse Chemistry Teaching Resources

Excess And Limiting Reagents Chemistry Libretexts
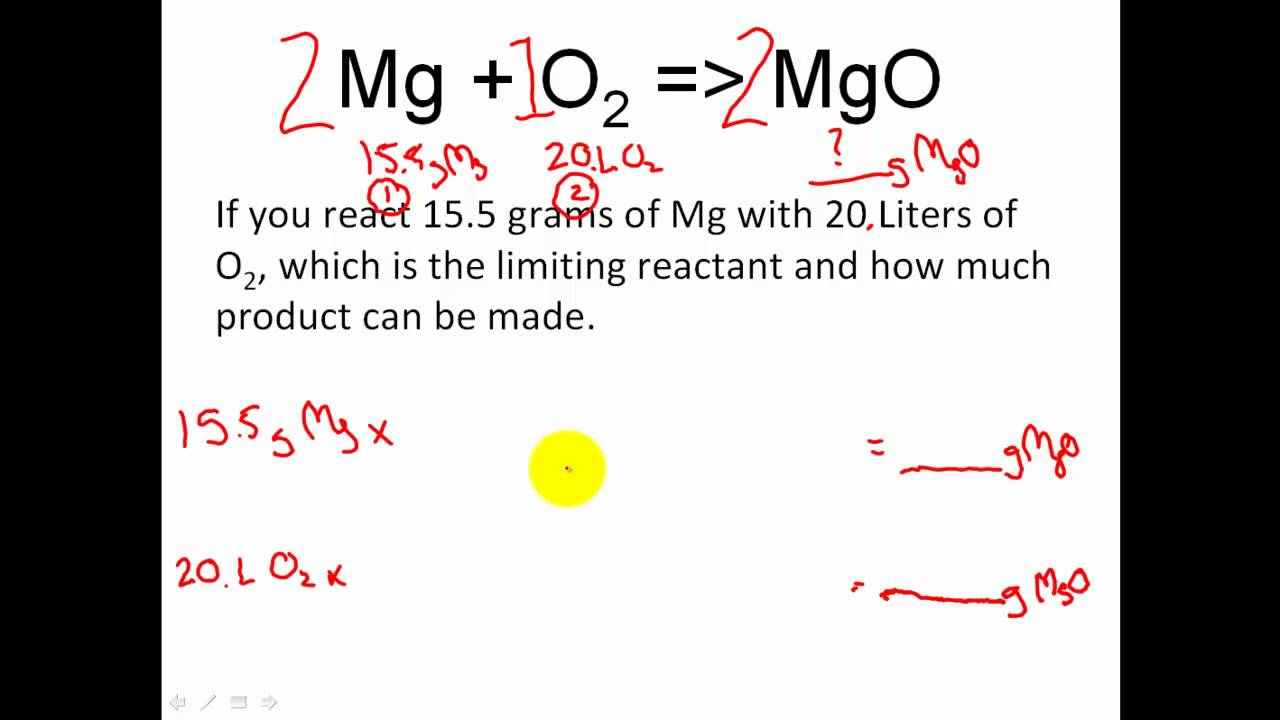
Stoichiometry Limiting Reactant Excess Reactant Stoichiometry Moles Youtube