Elements that have similar properties are arranged in groups or families - vertical columns. Metals In The Periodic Table So because most elements of the Table are metals it makes sense to begin by looking at them.

The Parts Of The Periodic Table
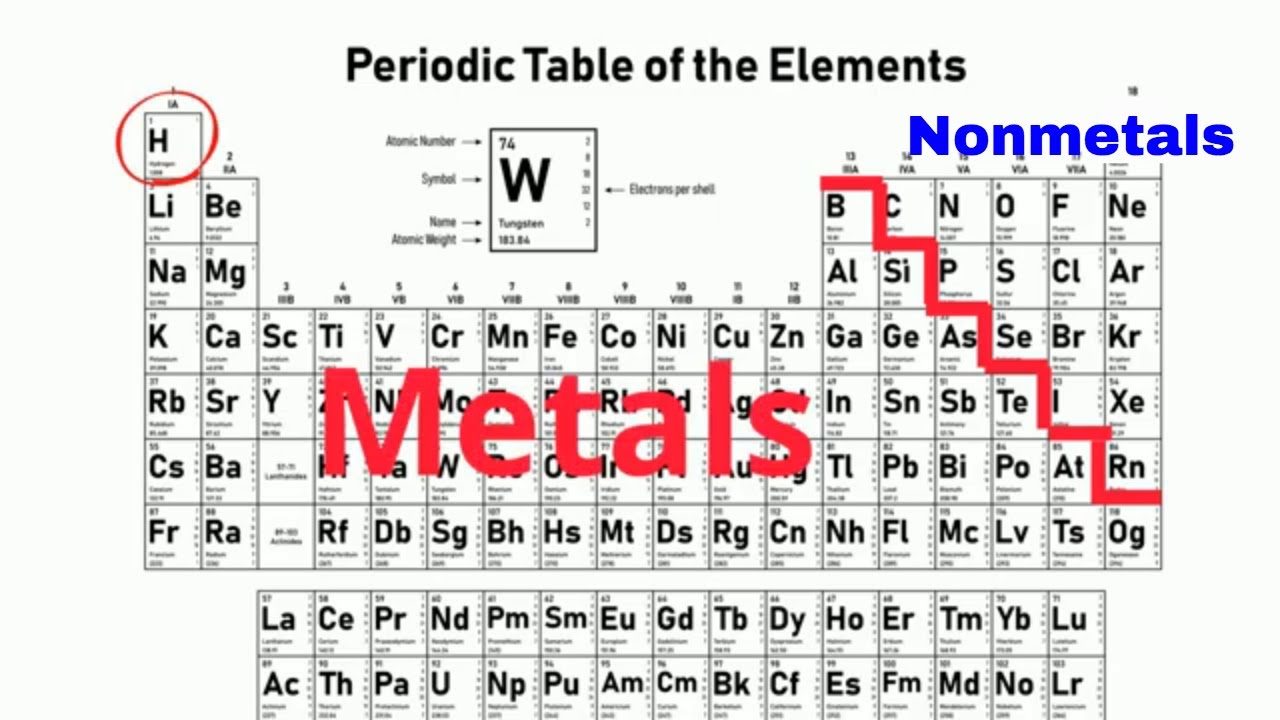
They are separated by a diagonal band of semimetals.
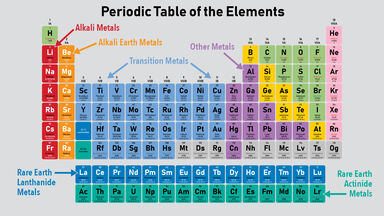
Where are the metals in the periodic table. The alkaline earth metals are found in column 2 on the left side of the Periodic Table. The metals consist of the alkali metals alkaline earths transition metals lanthanides and actinides. The three broad categories of elements are metals nonmetals and metalloids.
In chemistry the elements which are usually considered to be metals under ordinary conditions are shown in yellow on the periodic table below. They are grouped together in the middle to the left-hand side of the periodic table. Look up chemical element names symbols atomic masses and other properties visualize trends or even test your elements knowledge by playing a periodic table game.
This line is often referred to as the staircase because of its shape. The six alkaline earth metals are. Periodic table of the elements materials science and academic information elements and advanced materials data scientific presentations and all pages designs concepts logos and color schemes herein are the copyrighted proprietary rights and intellectual property of American Elements.
Interactive periodic table showing names electrons and oxidation states. The periodic table also known as the periodic table of elements is organized so scientists can quickly discern the properties of individual elements such as their mass electron number electron configuration and their unique chemical properties. Interactive periodic table with up-to-date element property data collected from authoritative sources.
Atomic number of an element is equal to the number of protons inside the nucleus of its atom. The remaining elements are either metalloids B Si Ge As Sb and Te being commonly recognised as such or nonmetals. Periodic table in chemistry the organized array of all the chemical elements in order of increasing atomic number.
The general features of the long form periodic table are. There are in all 18 vertical columns and 18 groups in the long form periodic table. It is arranged according to the periodic law.
The horizontal rows are called. Atoms to the left of the. Most elements can be considered metals.
Our current periodic table lists 109 elements. Elements are arranged in order of increasing atomic number their physical and chemical properties show a periodic pattern. Get essential facts about the first 20 elements all in one convenient place including the name atomic number atomic mass element symbol group and electron configurationIf you need detailed facts about these elements or any of the higher numbered ones start with the clickable periodic table.
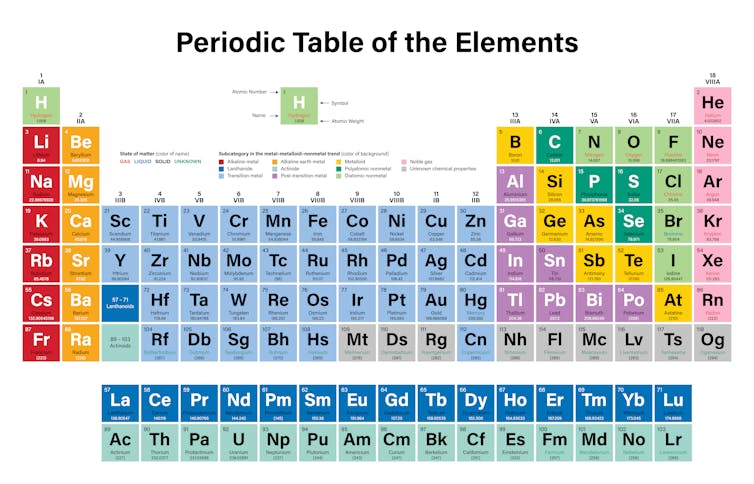
The periodic table also known as the periodic table of elements is a tabular display of the chemical elements which are arranged by atomic number electron configuration and recurring chemical propertiesThe structure of the table shows periodic trendsThe seven rows of the table called periods generally have metals on the left and nonmetals on the right. In the periodic table the vertical columns are called groups and the horizontal rows are called periods. Metals reside on the left side of the table while non-metals reside on the right.
Nonmetals are located on the righthand side of the periodic table. Except for Germanium Ge and Antimony Sb all the elements to the left of that line can be classified as metals. Metals on the Periodic Table The majority of elements on the periodic table are metals.
When the elements are thus arranged there is a recurring pattern called the periodic law in their properties in which elements in the same column group have similar properties. Metalloids have properties of both metals and nonmetals. They are generally harder and denser than alkali metals have 2 electrons in their outermost s sub-shell and each make a distinct color in their flames.
This periodic table groups elements according to type. In the periodic table you can see a stair-stepped line starting at Boron B atomic number 5 and going all the way down to Polonium Po atomic number 84. The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers.
Metals are lustrous good conductors of electricity and readily shaped they are ductile and malleable whereas solid nonmetals are generally brittle and poor electrical conductors. In the long form periodic table the elements are arranged in the order of their atomic numbers. If you look at the Periodic table you will find that the metal elements are located between atomic number 5 Boron B all the way to atomic number 84 Polonium Po.
Most periodic tables print a thick black line to show the division between metals and nonmetals. Metal blue nonmetal yellow or metalloid red. Metals are located on the left of the periodic table and nonmetals are located on the upper right.
Most elements are metals. Visualize trends 3D orbitals isotopes and mix compounds. The modern periodic table is based on the modern periodic law put forward by the English physicist Henry Moseley which states that the properties of.
How To Identify Metals Nonmetals And Metalloids On The Periodic Table Youtube
File Periodic Table Metals Svg Wikimedia Commons
Understanding The Periodic Table Through The Lens Of The Volatile Group I Metals