When a fluorine atom bonds to almost any other atom except another F atom the bond has ionic character because of partial transfer of an electron from the other atom to fluorine. The nearer the difference in electronegativity between atoms comes to zero the purer the covalent bond becomes and the less polarity it has.
How Does Electronegativity Affect A Chemical Bond Socratic
Electronegativity is a measure of the attraction of an atom for the electrons in a chemical bond.
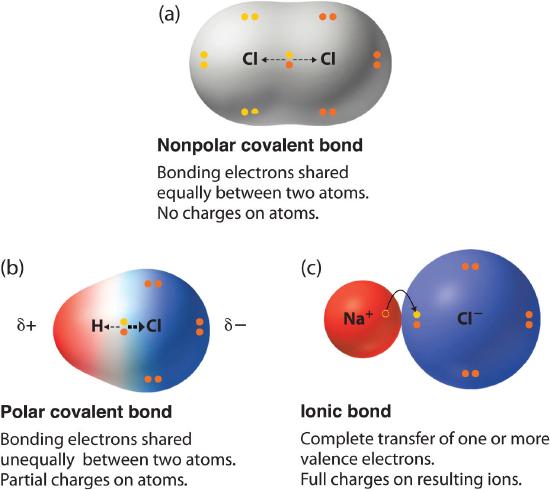
How does electronegativity affect bonding. The electron density that comprises the covalent bond is located halfway between the two atoms. When two atoms combine the difference between their electronegativities is an indication of the type of bond that will form. The more electronegative an atom is the more its bonds to other atoms will have ionic character especially to elements that have metallic character.
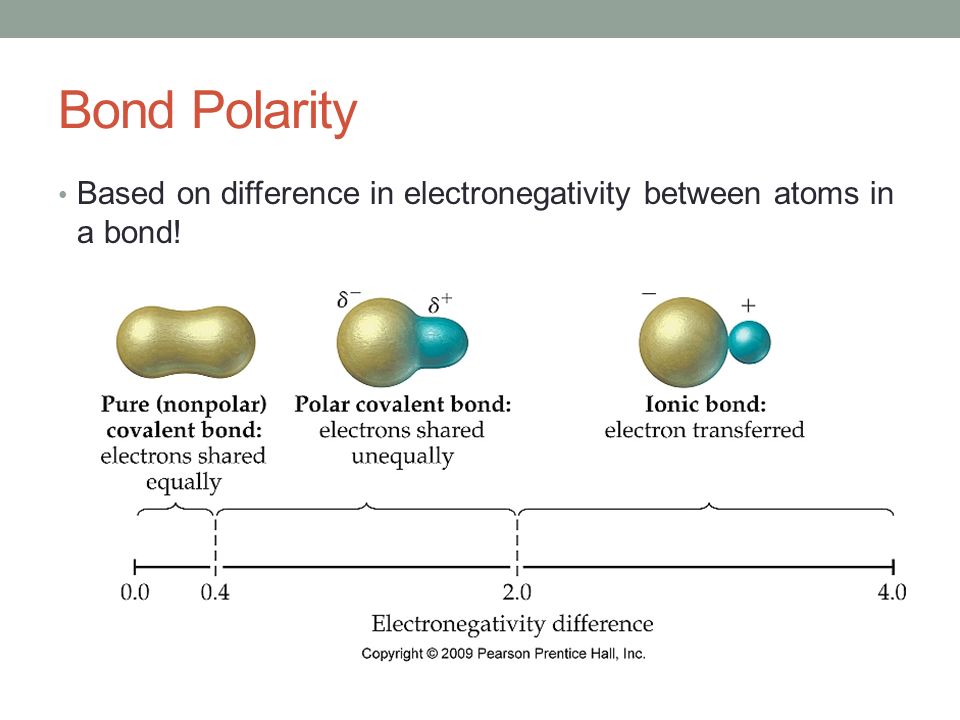
In a simple molecule like HCl if the bond is polar so also is the whole molecule. A large electronegativity difference leads to an ionic bond. Electronegativity and Bond Type The absolute value of the difference in electronegativity ΔEN of two bonded atoms provides a rough measure of the polarity to be expected in the bond and thus the bond type.
Electronegativity is the strength an atom has to attract a bonding pair of electrons to itself. The ability of an atom in a molecule to attract shared electrons is called electronegativity. The most strongly electronegative atom is fluorine.
The higher the electronegativity of an atom the greater its attraction for bonding electrons. Ionic bonding being a chemical bond gives rise to ions that are opposite charged in nature. In part b Ca has a low electronegativity.
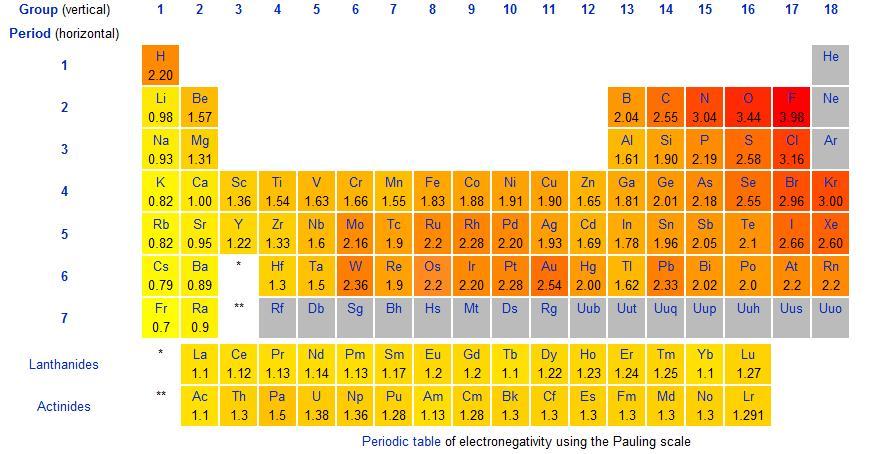
As a rule an electronegativity difference of 2 or more on the Pauling scale between atoms leads to the formation of an ionic bond. The chart shows electronegativities from sodium to chlorine ignoring argon since it does not does not form bonds. In VSEPR theory electronegativity of atomsgroups will effect bond angles due to changes in the distribution of electron pairs around the central atom and thus changes in severity of electron pair repulsion.
The electronegativity affects the chemical bond in the following. If the difference between the electronegativities of the two atoms is small neither atom can take the shared electrons completely away from the other atom and the bond will be covalent. A difference of less than 2 between atoms leads to covalent bond formation.
The most electronegative atom is fluorine F. A small electronegativity difference leads to a polar covalent bond. This effect only holds true for a row in the periodic table because the attraction between charges falls off rapidly with distance.
During the ionic bonding metals electrons are lost for positive ion formation and the non-metals. The compound with the greatest difference in electronegativity between the atoms in the compound is the most polar and therefore most soluble compound. No electronegativity difference between two atoms leads to a pure non-polar covalent bond.
The electronegativity depends upon the nature of the electronic configuration. The electronegativity of the central atom does not really influence bond angles in any meaningful way. When the difference is very small or zero the bond is covalent and nonpolar.
When two atoms combine the difference between their electronegativities is an indication of the type of bond that will form. Since O is more electronegative than S CaO is more polar and therefore is more readily dissolved in water. When a chlorine atom covalently bonds to another chlorine atom the shared electron pair is shared equally.
Across a period from left to right the electronegativity of atoms increases. The ability of an atom in a molecule to attract shared electrons is called electronegativity. Polar bonds and polar molecules.
When it is large the bond is polar covalent or ionic. As you move from left to right. Electronegativity symbol χ measures the tendency of an atom to attract a shared pair of electrons or electron density.
The results also show that electronegativity is a major influence on covalent bond lengths and the set of electronegativity scale and covalent radii proposed in this work can be used to calculate covalent bond lengths in different environments that have not yet been experimentally measured. Electronegativity is a measure of an atoms attraction for the electrons in a bond. The size of atoms and the orbitals used and useable are much more important.
An atoms electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. Therefore electronegativity increases from left to right in a row in the periodic table.

Ppt 2 1 Polar Covalent Bonds Electronegativity Powerpoint Presentation Id 2237583
Polarity How Does Electronegativity Affect The Polarity Of A Covalent Bond How Does The Shape Of A Molecule Affect The Polarity Of A Molecule Ppt Video Online Download

Ppt Bond Polarity And Electronegativity Powerpoint Presentation Free Download Id 5712777
8 4 Bond Polarity And Electronegativity Chemistry Libretexts